A simple, efficient, user-friendly adjudication software to connect
the key adjudication actors and manage online adjudication operations.
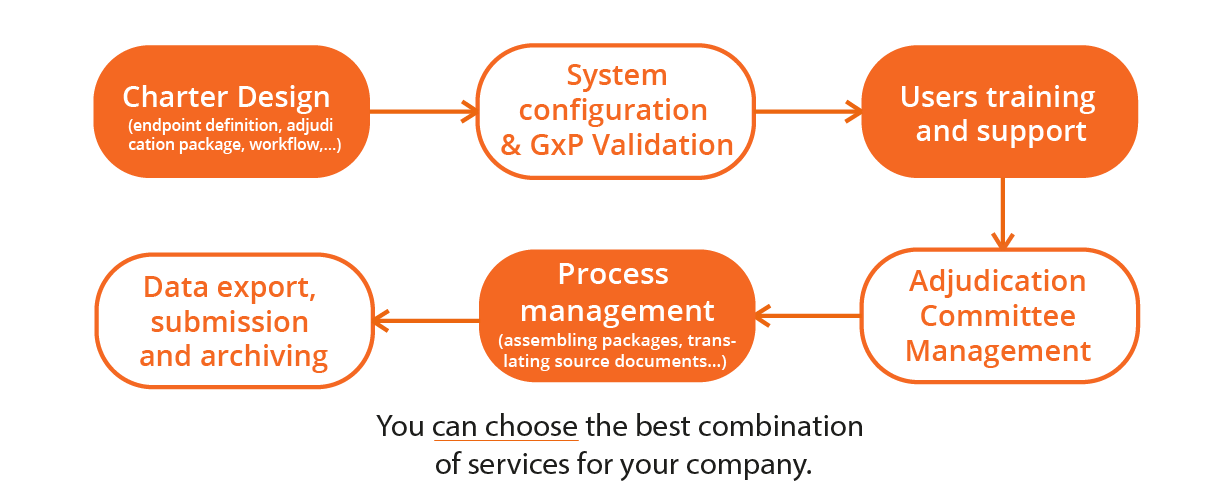
To save money and time optimizing the set-up of multiple adjudication studies with different charter specifications.

eAdjudication® provides you with an adjudication software platform dedicated to you, physically separated from other customers. It can be configured to exactly match your Standard Procedures (SOPs) while retaining the flexibility of using it for many different studies, with their specific forms, workflows, users' roles, custom reports, and exports.

You can match your SOPs with the flexibility required to serve different sponsors or studies. You gain all the possible economies of scale of using a single software platform for all of your studies.
KNOW MORETo support specific processes & workflows for event management, assessment, disagreement detection and resolution.

eAdjudication® flexible delivery model has been designed to comply with any procedures that each Sponsor decides to implement. Different algorithms for event assignment to reviewers; two, three or more levels for panel assessments; any workflow chosen for disagreement management, detection and resolution; all are efficiently handled by Ethical's engineers and delivered within short timeline.

With a single platform, you can manage the most heterogeneous adjudication studies without training your staff on complex system configurations.
KNOW MOREIntegrate or import data into the platform, submit documents of various formats to adjudicators, receive committee members’ assessments, control disagreement rates.

eAdjudication® can connect to most EDC systems (RAVE, OPEN CLINICA, MEDRIO, REDCap, CASTOR, ORACLE CLINICAL…), and import data from files as needed. The event package accepts most of the common formats such as WORD, PDF, DICOM and EXCEL. Submission can be automated ensuring a balanced repartition per reviewer or pair of reviewers. Quality metrics are automatically collected throughout the study duration including disagreement rates. A process is available to import “clone events” (usually 5 or 10% of total events) to be re-submitted to the panel for Intra-Variability control.

Event preparation and processing is made easy for the sponsor team. You can report real time Quality Control metrics to your Managers and Study Sponsor without expensive data processing outside the platform.
KNOW MORETo present packages of documents of various formats (PDF, DICOM…) to committee members, all previously redacted and readily available for review.

eAdjudication® is fitted with a convenient, easily accessible DICOM viewer that offers a full range of tools for visualizing and managing the images. PDFs can be redacted manually. Image metadata can be redacted either automatically with Artificial Intelligence algorithm or manually.

Study Operations can start immediately after the Software delivery without complex staff trainings. Committee will deal with quality designed Adjudication Forms that can be saved and repurposed for future studies.
KNOW MORETo collect reviewers' assessments efficiently and in compliance with the charter – Reduce training and committee members’ errors.

Adjudication forms design, field labels, edit checks and data variables used to store the Adjudication Committee assessments are designed by the experienced Ethical's Staff to match exactly each Study Charter.

Study operations can start immediately after the software delivery without complex staff trainings. Committee will deal with quality designed Adjudication Forms that can be saved and repurposed for future studies.
KNOW MORETo collect event-related source medical records from sites in a timely and quality controlled manner – To track and manage queries’ resolution.

eAdjudication® provides Study Investigators with access to a dedicated area with the check-list of the medical records they need to upload. Source documents are tracked and versioned. The central staff is notified of new versions. All queries for a given site are managed, tracked and solved in a single place.

Your staff manages sites, medical records and queries in a timely and efficient manner. You can prevent one of the most common cause of delays in study completion.
KNOW MORETo ensure a timely and compliant execution of your adjudication studies by detecting operational bottlenecks and correcting them as fast as possible.

We have experience from dozens of studies and we understand the metrics that study staff need for monitoring and reporting. Events assessed, reviewers' workload, alerts for outliers and delays against operational benchmarks, events in each workflow status. Each represented by a dedicated widget that can be printed or exported.

Operations oversight and reporting to your managers or sponsor can't be faster and easier. All operational metrics are available in a single, customizable, view.
KNOW MORETo provide sponsors’ data managers with needed data exports during the study and to support event data submission at the end of the study.

eAdjudication® provides data managers with real-time self service data exports. Standard Exports include CSV, XML, Excel files with audit trail, events lifecycle history, adjudication outcomes, etc. Other customized formats (e.g. SAS) are available on request. A dedicated set of data and documents, suitable for submission to Authorities, is made available at the end of each study.

Adjudication data are always available in real-time as soon as the study starts. The self-service export function is configured to match your specifications without complex or time-consuming operations.
KNOW MOREeAdjudication® offers such flexibility that the software configuration and support provision are tailored to exactly match your need.